‘Simple yet powerful’: Seeing cell secretion like never before
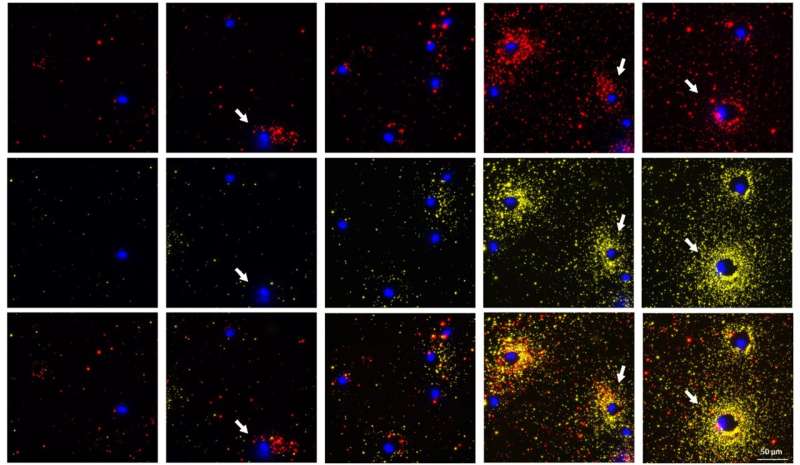
We have recently witnessed the stunning images of distant galaxies revealed by the James Webb telescope, which were previously visible only as blurry spots. Washington University in St. Louis researchers have developed a novel method for visualizing the proteins secreted by cells with stunning resolution, making it the James Webb version for visualizing single cell protein secretion.
The researchers, led by Srikanth Singamaneni, the Lilyan & E. Lisle Hughes Professor of Mechanical Engineering & Materials Science in the McKelvey School of Engineering, and Anushree Seth, a former postdoctoral scholar in Singamaneni’s lab, developed the FluoroDOT assay, which they introduced in a paper on August 5, 2022 in the journal Cell Reports Methods. The highly sensitive assay is able to see and measure proteins secreted by a single cell in about 30 minutes.
In collaboration with researchers in the Washington University School of Medicine and other universities, they found that the FluoroDOT assay is versatile, low-cost and adaptable to any laboratory setting and has the potential to provide a more comprehensive look at these proteins than the widely used existing assays. Biomedical researchers look to these secreted proteins for information on cell-to-cell communication, cell signaling, activation and inflammation, among other actions, but existing methods are limited in sensitivity and can take up to 24 hours to process.
What makes the FluoroDOT assay different from existing assays is that it uses a plasmonic-fluor, a plasmon-enhanced nanolabel developed in Singamaneni’s lab that is 16,000 times brighter than conventional fluorescence labels and has a signal-to-noise ratio of nearly 30 times higher.
“Plasmonic-fluors are composed of metal nanoparticles that serve as antenna to pull in the light and enhance the fluorescence emission of molecular fluorophores, thus making it an ultrabright nanoparticle,” Singamaneni said.
This ultrabright emission of plasmonic-fluor allows the user to see extremely small quantities of secreted protein, which they are unable to do in existing assays, and measure the high-resolution signals digitally using the number of particles, or dot pattern, per cluster, or spot, using a custom-built algorithm. In addition, it doesn’t require special equipment. Singamaneni and his collaborators first published their work with the plasmonic-fluor in Nature Biomedical Engineering in 2020.
The patent-pending plasmonic fluor technology is licensed by the Office of Technology Management at Washington University in St. Louis to Auragent Bioscience LLC.
“Using a simple fluorescence microscope, we are able to simultaneously image a cell along with the spatial distribution of the proteins secreted around it,” said Seth, who worked on this project as a postdoctoral scholar in Singamaneni’s lab and continues to work on it as a principal scientist (cellular applications) for Auragent Bioscience. “We saw interesting secretion patterns for different cell types. This assay also enables concurrent visualization of two types of proteins from individual cells. When the multiple cells are subjected to the same stimuli, we can distinguish the cells that are secreting two proteins at the same time from the ones that are only secreting one protein or are not secreting at all.”
To validate the technology, the team used proteins secreted from both human and mouse cells, including immune cells infected with Mycobacterium tuberculosis.
One of the collaborators and co-authors, Jennifer A. Philips, MD, Ph.D., the Theodore and Bertha Bryan Professor in the departments of Medicine and Molecular Microbiology and co-director of the Division of Infectious Diseases in the School of Medicine, has used the FluoroDOT assay in her lab.
“When Mycobacterium tuberculosis infects immune cells, those cells respond by secreting important immune proteins, called cytokines,” Philips said. “But not all cells respond to infection the same way. The FluoroDOT assay allowed us to see how individual cells in a population respond to infection—to see which cells are secreting and in which direction. This was not possible with the older technology.”
Team develops rapid SARS-CoV-2 test based on new plasmonic-fluor biolabeling technology
Anushree Seth et al, High-resolution imaging of protein secretion at the single-cell level using plasmon-enhanced FluoroDOT assay, Cell Reports Methods (2022). DOI: 10.1016/j.crmeth.2022.100267
Citation:
‘Simple yet powerful’: Seeing cell secretion like never before (2022, August 5)
retrieved 5 August 2022
from https://phys.org/news/2022-08-simple-powerful-cell-secretion.html
This document is subject to copyright. Apart from any fair dealing for the purpose of private study or research, no
part may be reproduced without the written permission. The content is provided for information purposes only.
For all the latest Science News Click Here
For the latest news and updates, follow us on Google News.

