Engineering nitrogenases for synthetic nitrogen fixation: From pathway engineering to directed evolution
Fertilizers are one of the main reasons that we are able to grow enough crops to feed the almost 8 billion humans living on Earth. Modern agriculture depends largely on nitrogen-based fertilizers, which significantly increase the yield of crops. Unfortunately, a great portion of these fertilizers are produced at an industrial level, consuming fossil fuel energy and causing nitrogen pollution.
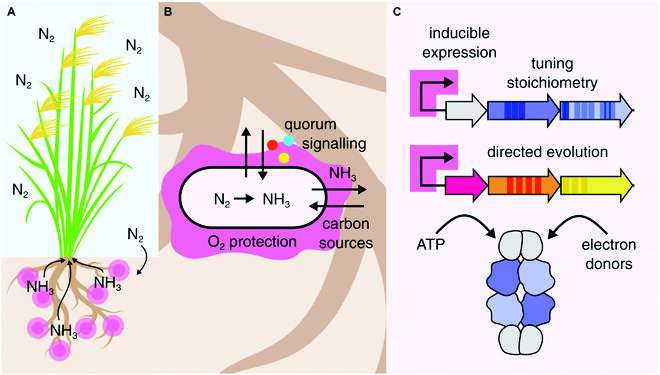
One attractive way to minimize our use of industrially produced fertilizers is to harness the power of nitrogenases. These enzymes, found in various microorganisms, can carry out nitrogen fixation, i.e., the conversion of nitrogen gas (N2) to ammonia (NH3), which is a more biologically useful form for plants.
Using bioengineering tools, we could introduce the genes that code for nitrogenases from one species of bacteria into another species (a ‘heterologous host’). The ultimate goal of this process would be to engineer a new type of bacteria that can survive in the field alongside crops and provide plants with NH3, thereby reducing the need for so much industrial fertilizer.
However, this strategy has proven to be difficult to pull off, and researchers are still trying to overcome many roadblocks. Motivated by this, a team of scientists including Professor Mark Isalan from Imperial College London, UK, recently published a review article in BioDesign Research. In it, they highlight recent progress towards understanding and surpassing the challenges found while engineering organisms to express and use nitrogenase.
Nitrogenase and its associated enzymes are coded by nif gene clusters, which are present across various species of bacteria with some variations. Though these gene clusters are complex, some researchers have managed to pinpoint a minimal set of genes that are essential for nitrogen fixation. This is essential knowledge for the logical next step: transferring custom nif genes to a heterologous host.
One of the main issues in engineering nitrogen-fixating bacteria is making sure the transferred nif clusters are compatible with the survival of the organism. As explained in the review, producing nitrogenases and using them is a resource- and energy-intensive endeavor for bacteria. If the engineered organisms derive no benefit from expressing the new genes, they are very likely to be outcompeted by other bacteria in the soil or escape the ‘useless’ genes by evolving against them.
Moreover, the expression of nitrogenase is usually regulated by complex chemical pathways that, in turn, are regulated by environmental variables, including nitrogen concentration or chemical signaling from plants. Thus, many approaches for increasing NH3 production in heterologous hosts haven’t led to much success. Another thing to keep in mind is that nitrogenases are easily damaged by oxygen, and the heterologous host will need mechanisms to avoid this issue.
The review puts great emphasis on symbiosis, as the survival of engineered organisms out in the field will depend on their relationship with the target crop. For starters, the crop plant should ‘reward’ nearby nitrogen-fixating organisms by exchanging sugars with them, as well as provide appropriate chemical signals to guide nitrogenase expression. In turn, the bacteria should ideally kick off NH3 production only when near the plant’s roots.
Hence, the overall goal of engineering organisms towards a symbiotic relationship is to give both species an edge in the fight for survival. However, as Isalan remarks, we are yet to achieve this target: “Survival of engineered bacteria in real-world environments has not yet been explored in detail, and much work needs to be done toward engineering synthetic symbioses that survive competition with other soil microorganisms.”
The article also introduces a promising approach called ‘directed evolution,’ which consists of controlled laboratory experiments where engineered bacteria are made to compete with one another. The experiments are set up so that the ‘winning’ genes are more suitable in a real environment or, alternatively, shed some light on specific molecular mechanisms of nitrogen fixation.
There are certainly many hurdles to overcome if we want engineered bacteria to help us fertilize the soil, and there are even more possible ways in which we may tackle these challenges. “Whichever approach is used, improving nitrogenase activity toward nitrogen-fixing plants is a complex and challenging engineering problem, which will require creative solutions as varied and dynamic as those we see in the natural world,” concludes Isalan.
More information:
Emily M. Bennett et al, Engineering Nitrogenases for Synthetic Nitrogen Fixation: From Pathway Engineering to Directed Evolution, BioDesign Research (2023). DOI: 10.34133/bdr.0005
Citation:
Engineering nitrogenases for synthetic nitrogen fixation: From pathway engineering to directed evolution (2023, March 8)
retrieved 8 March 2023
from https://phys.org/news/2023-03-nitrogenases-synthetic-nitrogen-fixation-pathway.html
This document is subject to copyright. Apart from any fair dealing for the purpose of private study or research, no
part may be reproduced without the written permission. The content is provided for information purposes only.
For all the latest Science News Click Here
For the latest news and updates, follow us on Google News.

